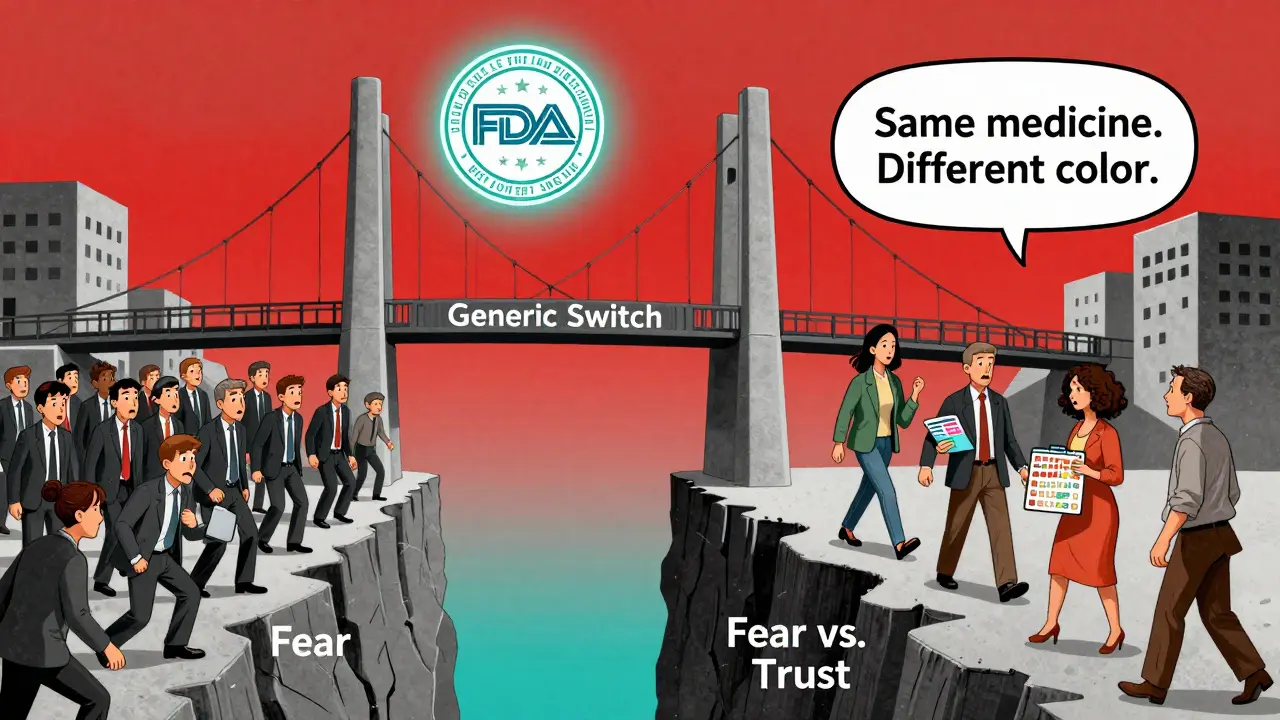
When you switch from a brand-name drug to a generic version, your body doesn’t change - but your perception might. And that perception can make or break whether you take your medicine as prescribed.
Over 90% of prescriptions filled in the U.S. are for generic drugs. They cost 80-85% less than brand-name versions. For many, that’s life-changing. But here’s the catch: after switching, adherence - meaning taking the right dose at the right time - drops by an average of 15%. For some medications, it drops even more. And it’s not because generics are weaker. It’s because of what’s happening in your mind.
Why Do People Stop Taking Their Generic Meds?
It’s not science. It’s psychology.
A 2018 study in the European Journal of Public Health gave people identical placebo pills - one labeled as a brand-name drug, the other as generic. The group with the generic label reported 18.3% more pain, took 31.6% more over-the-counter pills, and were 23.7% more likely to stop taking the pill before the week was over. Same pill. Same dose. Different label. Different outcome.
This is called the nocebo effect - the opposite of placebo. When you believe something won’t work, your body starts to act like it doesn’t. And in the case of generics, that belief is often fueled by:
- Changed pill shape, color, or markings
- Confusing packaging
- Stories from friends or online forums
- Doctors or pharmacists not explaining the switch
One patient on Reddit wrote: “I’ve been on Lamictal for 12 years. When they switched me to generic, I started having seizures. I went back to the brand - and they stopped.” The FDA later confirmed the generic met all bioequivalence standards. But the patient’s brain had already decided: this isn’t the same.
Which Medications Are Most Affected?
Not all drugs are created equal when it comes to switching. Some are more sensitive to even tiny changes.
High-risk categories:
- Levothyroxine (for thyroid): A 2017 NEJM study found a 0.8% higher hospitalization rate after switching. Why? Thyroid levels need to be extremely stable. Even a 5% difference in absorption can throw off your metabolism.
- Warfarin (blood thinner): Small changes in how fast the drug is absorbed can lead to clots or bleeding. Pharmacists often recommend sticking with the same manufacturer.
- Antiepileptics (like lamotrigine): Seizure control is fragile. A 2021 lawsuit against Teva involved over 8,000 patients who reported breakthrough seizures after switching. The FDA found no bioequivalence issue - but the fear stuck.
- Antidepressants: A 2020 study in CNS Drugs found 19.4% higher discontinuation rates after switching. Mood disorders are sensitive to subtle shifts in brain chemistry.
Low-risk categories:
- Statins (cholesterol): Adherence actually improved by 8.7% after switching - because copays dropped. People took their pills more consistently.
- Diabetes meds (like metformin): No significant difference in adherence. Most patients don’t notice a change.
The lesson? Don’t treat all generics the same. Some switches are safe. Others need care.
What’s Really Different Between Brand and Generic?
The FDA requires generics to have the same active ingredient, strength, dosage form, and route of administration as the brand. That’s the law. And bioequivalence testing? It’s strict: absorption must fall between 80-125% of the brand’s levels.
So what’s different?
- Inactive ingredients: Fillers, dyes, coatings. These can cause allergic reactions in 3.7% of people (per JAMA Dermatology). If you’re sensitive to lactose or red dye, check the label.
- Pill appearance: Color, shape, imprint. A 2020 University of Michigan study found 28.4% of elderly patients got confused - mixing up pills or skipping doses because they looked “wrong.”
- Manufacturers: The same generic drug can be made by different companies. One might use a different coating that changes how fast it dissolves. That’s why some patients do better sticking with the same manufacturer.
Here’s the truth: every brand-name drug varies slightly from batch to batch. The FDA allows up to 20% variation in absorption between different batches of the same brand. So if your brand drug changed over time, why should you panic when the generic does?

Best Practices for Patients
If you’re being switched to a generic, here’s what you can do:
- Ask your pharmacist: “Is this the same as my old pill?” They can show you the difference in shape or color. Seeing it helps.
- Use a pill organizer: If the new pill looks unfamiliar, a daily compartmentalized box helps you confirm you’re taking the right one.
- Don’t stop cold: If you feel weird, don’t quit. Call your provider. A headache or nausea might be temporary - or unrelated.
- Request the same manufacturer: Especially for thyroid, seizure, or blood-thinner meds. Ask: “Can I get the same generic brand as before?”
- Track your symptoms: Keep a simple log: “Day 1: no side effects. Day 3: mild headache. Day 5: back to normal.” Patterns help your doctor decide if it’s real or psychological.
One patient in a 2023 Kaiser Permanente study said: “I started using a pill box. I realized I was taking the same thing - just a different color. I felt silly. But I’ve been on it for 6 months now - no issues.”
Best Practices for Providers
Doctors and pharmacists have a huge role here. Too often, the switch happens silently - no explanation, no follow-up.
The American Pharmacists Association recommends a minimum 3.7-minute conversation during a switch. But in real life, only 19% of patients are even asked if they have concerns.
Here’s what works:
- Explain bioequivalence: “The FDA requires this generic to work the same way as your brand. It’s tested in thousands of people.”
- Describe the visual change: “This pill is now white instead of blue. It’s the same medicine - just made by a different company.”
- Highlight cost savings: “You’re saving $40 a month. That’s $480 a year.” For many, that’s worth it.
- Invite questions: “What worries you about this change?” Don’t assume they’re fine.
Pharmacists who call patients within 72 hours of a switch see a 31% increase in adherence. That’s not magic. It’s care.
Technology Is Helping - But Not Enough
Electronic prescribing systems now flag brand-to-generic switches and alert pharmacists. One study showed this led to a 22.4% rise in counseling.
Some pharmacies now use medication synchronization - aligning all refill dates. That helped generic users stay on track 18.3% better.
And new AI tools like AiCure use smartphone cameras to verify patients are taking their pills. In a 2023 study, adherence during generic transitions jumped 37% with this system.
But tech alone won’t fix the problem. If a patient doesn’t trust the medicine, no app will help.
The Bigger Picture: Cost vs. Confidence
Generic drugs save the U.S. healthcare system $1.1 trillion a year. That’s real. But if patients stop taking them, those savings vanish - replaced by ER visits, hospitalizations, and worsening disease.
Low-income patients often benefit the most from generics - their adherence improves because they can afford them. But high-income patients? They’re more likely to refuse generics out of distrust. A 2021 Consumer Reports survey found 62% of Americans trust brand-name drugs more - even when the generic costs 80% less.
The FDA and drugmakers are starting to respond. Starting January 1, 2025, generic manufacturers must include transition-specific patient education materials. And the Generic Pharmaceutical Association is launching a “Seamless Switch” certification - requiring consistent pill appearance for critical drugs.
But the biggest fix? Communication. Not regulation. Not technology. Talking.
What Should You Do Today?
If you’re switching to a generic:
- Don’t panic.
- Ask your pharmacist: “Is this the same medicine?”
- Use a pill organizer.
- Track how you feel - for 2 weeks.
- If something feels off, call your doctor - don’t quit cold.
If you’re a provider:
- Don’t assume patients understand the switch.
- Take 5 minutes to explain it.
- Call them back in 72 hours.
- For high-risk drugs, consider sticking with one manufacturer.
Generics aren’t inferior. They’re identical - in active ingredients, in testing, in safety. But they’re not identical in perception. And that’s where the real work begins.
Are generic drugs as effective as brand-name drugs?
Yes - by law, generic drugs must meet the same FDA standards as brand-name drugs. They contain the same active ingredient, strength, dosage form, and route of administration. Bioequivalence testing ensures they are absorbed into the bloodstream at the same rate and extent. Over 90% of prescriptions in the U.S. are generic, and they’ve been safely used for decades. The difference isn’t in effectiveness - it’s in perception.
Why do I feel different after switching to a generic?
You’re likely experiencing the nocebo effect - where expecting a problem leads to real symptoms. Changes in pill color, size, or taste can trigger this. In one study, patients given identical placebos labeled as generic reported 18% more pain than those given the same pill labeled as brand. It’s not the drug - it’s your brain. Keep taking it for 1-2 weeks. If symptoms persist, talk to your provider.
Should I avoid switching for certain medications?
For drugs with a narrow therapeutic index - like levothyroxine, warfarin, or seizure medications - small changes in absorption can matter. Talk to your doctor before switching. Some patients do better staying with the same manufacturer or even the brand if cost allows. But for most medications - statins, blood pressure pills, diabetes drugs - switching is safe and often improves adherence because of lower cost.
Can I request to stay on the brand-name drug?
Yes. Your doctor can write “Dispense as Written” or “Do Not Substitute” on the prescription. Insurance may require prior authorization or higher copays. But if you’ve had trouble with generics before - or if you’re on a high-risk medication - you have the right to ask.
What if I can’t afford the brand-name drug anymore?
Don’t stop taking your medication. Talk to your pharmacist or doctor. Many manufacturers offer patient assistance programs. Some pharmacies have $4 generic lists. And under the 2024 Inflation Reduction Act, Medicare beneficiaries now pay no more than $2,000 a year out-of-pocket for prescriptions. You have options - but skipping doses isn’t one of them.


Adam Kleinberg
March 10, 2026 AT 12:13Let me get this straight - we’re telling people to trust a pill that looks different and costs less because some FDA bureaucrat says it’s ‘bioequivalent’? Please. The same companies that make the brand-name drugs also make the generics - just with cheaper fillers and dyes that trigger real reactions. I’ve seen people go from stable to seizing after a switch. And no, it’s not ‘nocebo’ - it’s corporate fraud wrapped in regulatory jargon. They don’t test long-term effects. They test absorption over 72 hours. That’s not medicine - it’s a gamble with people’s lives.
Gene Forte
March 11, 2026 AT 16:41Every great change in healthcare starts with a conversation. Switching to a generic isn’t about cutting costs - it’s about giving people access to life-saving medicine. The fear isn’t irrational, but it’s not unchangeable either. A simple 5-minute talk - showing the pill, explaining the science, listening to concerns - can rebuild trust. We don’t need more regulations. We need more humans showing up. And when we do? Adherence goes up. Lives improve. It’s not magic. It’s care.
Kenneth Zieden-Weber
March 12, 2026 AT 16:10So let me get this - you’re telling me that if I believe a white pill won’t work, my brain will trick my body into feeling sick… but if I believe a blue pill is magic, I’ll feel better? That’s not psychology. That’s a cult. And the FDA’s 80-125% absorption window? That’s like saying ‘your car’s engine is fine as long as it runs between 30% and 125% of its original power.’
Meanwhile, people with epilepsy are getting breakthrough seizures because a generic manufacturer changed the coating - and no one told them. This isn’t about perception. It’s about sloppy manufacturing and zero transparency. The system is broken. And we’re blaming patients for noticing.
Chris Bird
March 12, 2026 AT 22:26Generic drugs are scam. They use bad chemicals. People get sick. America lie. Nigeria use only brand. We not stupid.
David L. Thomas
March 13, 2026 AT 09:20Look - the nocebo effect is real, but so is the pharmacokinetic variability in generics, especially with narrow-therapeutic-index drugs. The FDA’s bioequivalence criteria are statistically sound, but they’re population-level averages. For an individual with CYP2C9 polymorphisms or GI motility issues, a 5% shift in AUC can be clinically significant.
And let’s not forget: multiple manufacturers = multiple excipient profiles. One company uses lactose monohydrate, another uses microcrystalline cellulose - and if you’re lactose intolerant? Congrats, you’ve got a pseudo-allergic reaction. This isn’t placebo. It’s a lack of granular labeling. We need batch-specific patient alerts in EHRs. Not just ‘generic approved.’
Shourya Tanay
March 13, 2026 AT 12:34As someone who’s managed chronic illness for over a decade, I’ve switched generics 4 times. The first time, I panicked. The second, I tracked symptoms in a journal. Third time? I asked my pharmacist to match the manufacturer. Fourth? I didn’t even notice.
It’s not about the pill. It’s about control. When you’re handed a new pill with no context, it feels like your body is being experimented on. But if you’re given agency - ‘Here’s what changed. Here’s how to monitor. Here’s when to call’ - then trust rebuilds.
Doctors don’t need to be pharmacists. But they need to know when to hand off the conversation. And pharmacists? They need time. Not just a checkbox.
LiV Beau
March 14, 2026 AT 03:20OMG YES. I switched to generic levothyroxine and felt like a zombie for 3 weeks 😩 I thought I was dying. Then I started using a pill box and wrote down how I felt each day - turns out the headache faded after 10 days. I’m so glad I didn’t quit! My endo said 70% of patients have this reaction. It’s wild how much our brains play tricks on us. 🙃
Also - if you’re switching, ask for the same manufacturer! Mine is Teva. Always Teva. 🤞
PS: My pharmacist called me 2 days later. That meant everything. ❤️
Denise Jordan
March 14, 2026 AT 20:07So let me get this: we’re spending billions to make people feel better about pills that are supposed to be the same? Why not just make all generics look like the brand? Or better yet - stop switching people without asking. This whole thing is a mess. I’m not paying $400 for a pill I can get for $5 if I’m just gonna stress about it. 🤷♀️